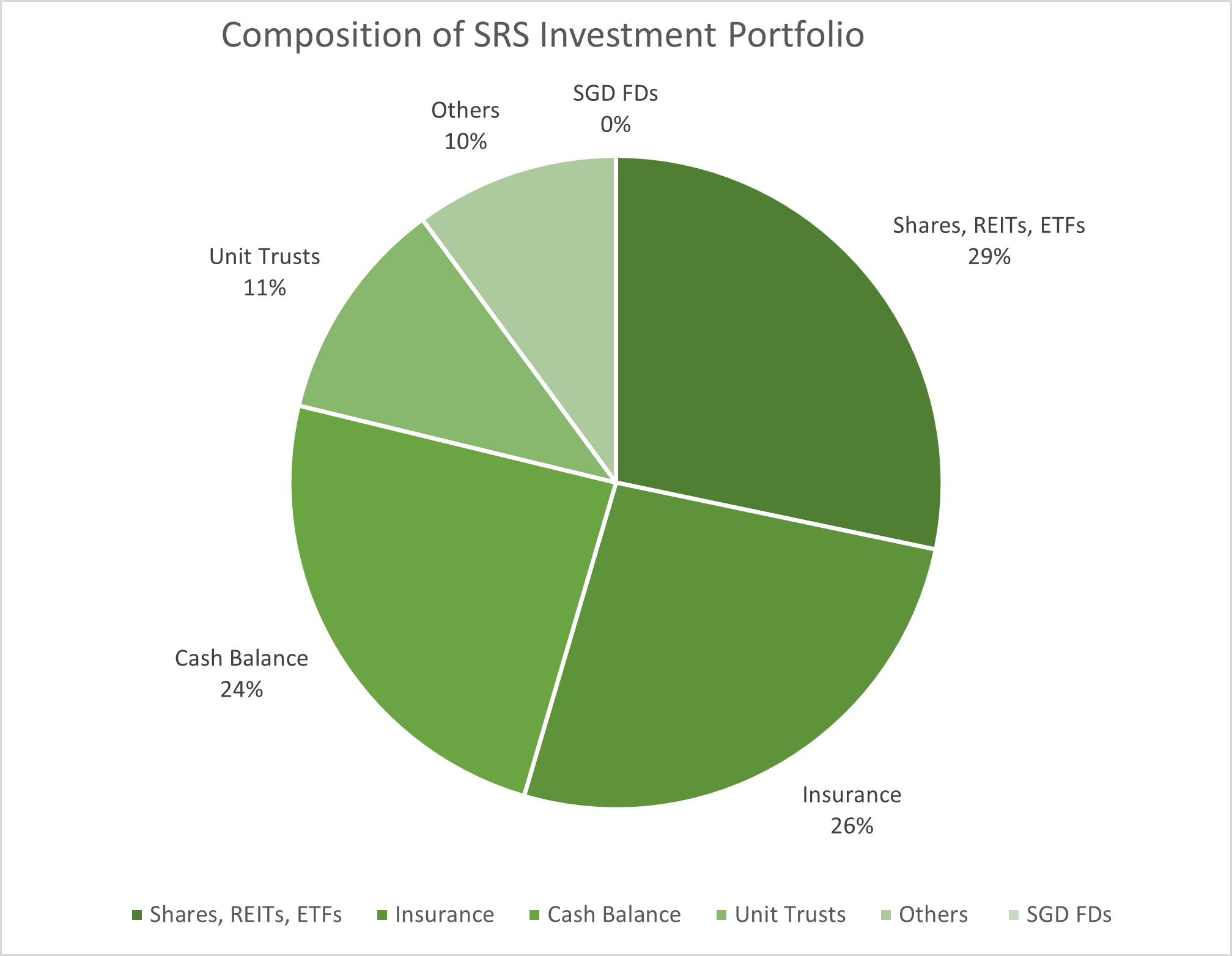
SRS Financials
ผู้เยี่ยมชม
30websiteproject@gmail.com
Trusted Guidance by SRS Financials (71 อ่าน)
13 ก.ย. 2568 05:30
Small cell lung cancer (SCLC) outcomes remain poor. Approximately, 40–60% of SCLC patients develop brain metastases (BMs) [1], [2], [3] and less than 15% survive beyond two years [4], [5]. In lung cancer management, contemporary advances in systemic therapies and focal radiation techniques have also tended to improve outcomes more in non-small cell lung cancer (NSCLC) than SCLC over the last decade [4]. For patients with brain metastases, whereas upfront SRS Financials has become the preferred treated for limited brain metastases arising from most solid tumor histologies [6], whole brain radiotherapy (WBRT) remains the standard of care for SCLC [7]. Although WBRT has demonstrated efficacy in controlling both local and distant intracranial disease there is now increasing concern about its acute and late toxicities particularly with respect to cognitive function and quality of life [8].
Stereotactic ablative radiation techniques such as radiosurgery (SRS) and fractionated stereotactic treatment (FSRT) are standard options for metastatic NSCLC and other solid tumor histologies with multiple randomized trials demonstrating similar overall survival and improvement in cognitive outcomes and quality of life (QoL) compared to WBRT . However, SCLC patients were excluded from these landmark trials due to multiple factors including concerns for short interval, multi-lesion central nervous system (CNS) progression specific to patients with SCLC histology, as well as the prevalent policy of prophylactic cranial irradiation (PCI) which has limited both the ability to study SRS without prior brain radiation and to randomize patients to arms that included WBRT , . Due to the paucity of data on SRS, WBRT has endured as the preferred treatment strategy for SCLC patients with one or more brain metastasis.

Today almost half of the world’s population is online and growing exponentially by 8% per year . Among these, over 2.8 billion people use social media. With nearly 79% of Twitter users outside the US , this modality offers a unique platform for global engagement. We have previously shown how this tool may be used to enable rapid dissemination of specialty-specific knowledge . To inspect the neglected condition of SCLC with BMs we used the global lens of the Radiation Oncology Journal Club (#RadOnc #JC) to critically review a large international cohort who received upfront SRS as an emerging option for patients (FIRE-SCLC).
This #RadOnc #JC occurred on Twitter™ from Saturday, June 21st 8AM to Sunday, June 22nd 4PM Central Standard Time (CST) in accordance with our usual protocol. The online journal club included limited-time open-access of the publication and an asynchronous online discussion followed by a live-hour to reduce barriers to participation, targeted online invitations to encourage diverse perspectives across disciplines and regions, moderation by a resident (IP) and diverse faculty (MK, RS, HS), and attendance by the study’s lead author (CR). Structured discussion topics (T1, T2, T3…) included an introduction for more general audiences then critical appraisal of the cohort study for application locally and globally by patients and providers. Natural evolution of these topics was allowed to draw out new lines of inquiry. Throughout, tweets were summarized using the social media content organization platform Wakelet for easier access to the knowledge-base generated by the discussion. Following the journal club, tweets were further reviewed, systematically organized into themes, and checked against existing literature to form the basis of the collated findings presented in this paper.
Over 80 individuals from 10 countries participated in the journal club with up to 120 participants responding to the anonymous polls (Table 1). These included radiation oncologists, medical oncologists, family physicians, residents, and a patient advocate. There was a total of 496 tweets, 205 retweets, 128 tweets with links and over 2.9 million impressions (Table 1). The Altmetric Attention Score, one composite measure of scholarly impact and online engagement with research output, was 227 (top 5%) as of July 24, 2020 . Thematic analysis resulted in four themes: limitations of the study, the changing role of PCI, emerging alternatives and adjunct treatments, and the impact on patients including informed consent. A slim majority of participants had already offered SRS as first-line treatment for SCLC BMs (50.6%, n = 77). Radiation oncologists mainly prompted the clinical consideration of upfront SRS (72%, 15.6% medical oncologists, 3.1% patients; n = 32). Locally ablative treatments were usually done by the central nervous system (CNS) team (46.7%, 25.8% by anyone, 24.2% by an SRS/FSRT specialist; 3.3% or the lung cancer team; n = 120).
Participant demographics from the June 2020 #RadOnc #JC. Active participants were defined as those who contributed to the discussion (i.e. excluding retweets or introductions). Unique tweets were total tweets minus retweets. Demographics only includes active participants.
Our global journal club discussion suggests that occasional use of upfront SRS (delivered as a single fraction or fractionated locally ablative therapy) for carefully selected cases of SCLC BMs in combination with MRI active surveillance is not uncommon among the #RadOnc community; however, patterns of care analyses suggest that upfront WBRT is used in the vast majority of SCLC cases , . Although consideration of upfront SRS is likely to be prompted more often by radiation oncologists, medical oncologists and patients are becoming increasingly aware of this potential option. The majority of participating radiation oncologists’ respondents indicated that their centers have a dedicated CNS team to perform SRS, which was followed by individual SRS specialists. In a minority of places, lung teams are involved in SRS. Earlier SRS studies for BMs were limited to treating 3 to 4 intracranial lesions. However, technological advancement, increasing expertise, and encouraging results have paved the way for trials evaluating treatment for a larger number of intracranial lesions . Additionally, the increased training in SRS and desire to decrease toxicity is helping to drive increased study and early adoption of SRS in this setting .
The First-Line Radiosurgery for Small Cell Lung Cancer (FIRE-SCLC) study is the largest analyses for upfront SRS (without prior PCI or WBRT) for SCLC BMs (n = 710). Compared to a WBRT cohort (n = 219) it concluded a similar median overall survival (mOS, 6.5 for upfront SRS vs 5.2 months for WBRT; p = .003) against a trade-off of shorter time to central nervous system progression (TTCP, 9.0 months vs not reached for WBRT; hazard ratio 0.38; p < .001). The trade-offs observed with SRS without WBRT in this study, including inferior CNS disease control without a decline in OS, appear similar to other histologies where upfront SRS is already well established by multiple randomized trials .
However, our journal club highlighted some of the significant barriers to recommending upfront SRS as a standard option at this time. Despite a trend in literature, regulatory bodies, and some practices to accept lower-level evidence as practice-changing, there are key limitations of non-randomized data. Although propensity score analyses can help mitigate confounding variables, they are not a substitute for randomization and cannot guarantee balanced confounders or statements of causality . In light of the clinical reality that SCLC patients treated with SRS would often be highly selected, even after propensity score matching it is likely that that biases from uncontrolled and unmeasured confounding would remain. Moreover, the rate of post-treatment MRIs differed between study groups. In the WBRT cohort they were recommended only in symptomatic patients after radiation and only 46% had at least one follow-up MRI. This increased to 89% in the SRS cohort and could have increased the differences in time to CNS progression (TTCP) observed between SRS and WBRT patients. The differences in follow up MRIs may have also allowed for more disease-altering salvage interventions among the SRS patients. Indications for modality of salvage therapy (16% salvage with WBRT and 33.5% with SRS in the upfront SRS cohort) were also not collected in the study. Lack of information on systemic therapies and the delivery of SRS largely in the pre-immunotherapy era (last year of treatment 2018 in FIRE-SCLC) could limit contemporary interpretations. Newer immune checkpoint inhibitors (ICIs) in combination with platinum-based chemotherapy have become a standard first-line treatment options for extensive stage-SCLC following the results of the IMpower 133 in 2018 and CASPIAN in 2019 , , , . However, most studies on ICIs have not included SCLC patients with untreated brain metastases.
Widespread use of upfront SRS is also limited by current practice patterns including standards of care, availability and accessibility. FIRE-SCLC omitted patients who received PCI, which is the standard of care for limited-stage SCLC and a standard treatment option in extensive-stage SCLC with global variation in utilization , , , , . The majority of patients in its upfront SRS group (540/710; 76%) were from Japan, which has the largest concentration of MRIs in the world (52 MRI units per million capita) and guidelines strongly recommending against PCI with near consensus from 34/35 of its subcommittee members , . For low-to-middle income countries (LMICs), while it may be an aspiration to opt for MRI surveillance this may not always be realistic. Although NCCN guidelines for Africa suggest both PCI and MRI surveillance as standard options , the latter will be hard to achieve when its populations have among the lowest levels of access (0.5 MRI units and 0.26 treatment units per million capita with less than 4% of its radiation needs covered). In many parts of the world the financial toxicity of upfront SRS and MRI active surveillance is the major limitation for patients and their healthcare systems who must prioritize their costs of care. Furthermore, it is important to realize the decreased sensitivity of a standard MRIs for detecting small BMs compared with high resolution thin slice, double or triple dose MRI . For others that allow either CT or MRI surveillance, given the significantly lower sensitivity for CT , it is unclear if brain CT surveillance is an effective strategy . Each of these issues can limit outcome comparisons between SRS vs. WBRT, the feasibility of trials, and applicability for practice while carrying risk of widening global disparities unless examined and deployed carefully.
The evolving role of more standard radiation options was also debated. PCI was as an exclusion criteria of FIRE-SCLC’s upfront SRS, and it could be argued that the encouraging outcomes with first line SRS without PCI offer an additional challenge to routine PCI administration. Although most participants still regarded PCI as the standard of care for LS and a standard treatment option for ES-SCLC , some participants were warmer to the idea of its omission. In support of PCI in LS-SCLC, a landmark meta-analysis of 987 patients across 7 randomized trials from 1977 to 1994 suggested a 5.4% OS benefit at 3 years with PCI over observation , but some participants felt that its older methods including imaging in the pre-MRI era may no longer apply in the era of brain MRI staging and surveillance . The landmark EORTC trial showed a benefit in OS for patients receiving PCI after a good response to systemic therapy whereas a recent Japanese trial by Takahashi et al. showed no survival difference. A key difference was the frequency of brain imaging. Most patients in the EORTC trial did not have any baseline brain imaging (63%) whereas all patients in Takahashi trial had baseline and surveillance MRIs every 3 months for year 1 and every 6 months for year 2. In the Japanese study, despite a reduction in CNS progression events (33% vs 59% at 1 year; P < .001) with PCI, no significant differences in OS were observed in the setting of MRI surveillance which may have allowed for earlier and more effective salvage therapy. However, the CNS control benefit observed with PCI in the Japanese study has also allowed PCI to remain a preferred option for some providers and healthcare systems. Management of the thorax where failures traditionally occur has also evolved. Jeremic et al. showed an improvement in survival with consolidative thoracic radiation treatment (TRT) for patients with ES-SCLC in an older era, and the more recent CREST phase 3 randomized trial showed an OS benefit with consolidative thoracic RT in a secondary analysis at 2-years. Subgroup analyses from the CREST trial also suggested improved outcomes with decreased disease burden (less than three metastases) and potential consideration of dose escalation in select cases . Similar to changing paradigms for other histologies and sites , , , identification of a select patient group to benefit from consolidative radiation in SCLC may suggest a relative oligometastatic state where a combination of aggressive local management of the thoracic disease with modern systemic therapy may yield better outcomes.
Another theme centered on alternatives to the traditional standard of WBRT as patients may be living longer and focus shifts to patient-centered care. Although only one patient advocate participated, many physicians held strong views on offering options to patients that minimize toxicity. Neurotoxicity from CNS directed radiation can be difficult to assess due to several factors such as sequelae of the intracranial disease, other treatments, and comorbidities. However, adverse effects on memory and quality-of-life have been described within the first year of patients receiving cranial irradiation with increased rates in older patients and those receiving higher doses , . Hippocampal-avoidance radiation treatment (HA-RT) has emerged as a standard option to preserve neurocognitive function without compromising intracranial failures in NSCLC . Although to this point hippocampal avoidance techniques have not been firmly established by trials specifically in SCLC patients, some participants already declared the demise of any conventional whole brain radiation delivery techniques and apply HA to both PCI and WBRT for SCLC when all metastatic lesions are away from the hippocampus. Hopefully the open question regarding the role of hippocampal avoidance in PCI will be settled by ongoing trials (Table 2). Another option that may improve both local and distant brain control could be adding a simultaneous integrated boost with WBRT, although recent trials have excluded SCLC . ICIs with CNS efficacy either alone or in combination with RT were also discussed, although existing data is mainly from preclinical and NSCLC studies and ongoing SCLC trials are limited (Table 2).
Pending Relevant Clinical Trials in ES-SCLC Globally.
Brain RT = WBRT or HA-WBRT (excluding PCI), TTCF = Time to cognitive failure, HVLT-R = Hopkins Verbal Learning Test – Revised, Brain RT = WBRT or HA-WBRT (excluding PCI), MTD = Maximum Tolerated Dose, IPFS = Intracranial Progression Free Survival, ORR = Objective Remission Rate, cTRT = Consolidative Thoracic Radiation Treatment, Ipi = Ipililumab, Nivo = Nivolumab, PDL1i = PD-L1 Inhibitor, Durva = Durvalumab, Atezo = Atezolizumab, Tetra = Tetraprizumab, Trem = Tremilimumab, Ola = Olaparib, Tala = Talazopari
The importance of patient-centered care in any decision-making was also discussed, especially for newer techniques with more uncertainty. Participants agreed that the risks and benefits of all available options in the context of their level of evidence should be discussed with patients when eliciting informed consent. This is especially true for HA brain radiation approaches, SRS, and MRI surveillance without PCI, even when these strategies are included in common guidelines . Where available, patients should be offered clinical trials to decrease this uncertainty. Less clear was the interpretation of the “best available evidence.” Some participants more strictly adhered to the standard options that were based on high levels of evidence than others.
This journal club highlighted the key findings and implications of the FIRE-SCLC study, a large cohort comparing SRS to WBRT that has brought much attention to the management of SCLC with BMs.
With the hope that improving therapeutic options will lead to better prognoses for SCLC patients, there is a growing interest in reducing toxicities including neurocognitive and financial costs of care to achieve better patient-centered outcomes. However, although the patient perspective was mentioned during the journal club, we did not have sufficient content to draw insights on how this may better be incorporated into clinical trials or shared decision-making. Moreover, while neurocognitive function was commonly mentioned, its specific components and the domains of quality of life were not discussed in detail. This should be an ongoing area of discussion, both online and off, to help ensure that the therapies we offer and outcomes we measure are of importance to all stakeholders.
Important limitations of this retrospective analysis will need to be addressed with prospective data prior to widespread use of upfront SRS for SCLC brain metastases. This includes the true differences in CNS control in SCLC patients, the competing risks of cognitive and QOL effects from WBRT vs higher rates of CNS progression with SRS, the confirmation that SRS is associated with comparable OS in the SCLC setting as well as exploring the true person-centered costs to assist informed decision-making for responsible capacity building and utilization. Driven by projects like FIRE-SCLC, a number of trials have been launched (NCT03297788 ; NCT003391362 ) or are in development (NRG CC-009: SRS vs HA-WBRT for 1-10 SCLC metastases) to evaluate first-line SRS in SCLC. Nevertheless, the NCCN SCLC guidelines, which consider WBRT the standard of care, were recently updated to acknowledge SRS as a potential treatment option in patients with small numbers of BMs . Overall, the fundamental question may be whether the trade-offs that helped to establish SRS alone without WBRT for other tumor histologies, including inferior CNS disease control against superior cognition and QOL with comparable OS, will hold true for SCLC where higher propensity for CNS dissemination and caution amidst resource limitations may impede its uptake. If a similar collection of trade-offs are observed both prospectively and for broader patient populations, the role of upfront SRS for SCLC BMs with ongoing close MRI surveillance is likely to expand.
Recently, there has been newfound interest for SCLC trials to guide patients and providers navigating these questions for SRS, omission of PCI and MRI surveillance, hippocampal avoidance, consolidative TRT, & immunotherapy (Table 2). As an important adjunct, we hope that online discussions on a global platform will continue to foster a community of open scientific inquiry and clinical trial participation.
139.135.55.4
SRS Financials
ผู้เยี่ยมชม
30websiteproject@gmail.com